It will be harder to breathe in the mountains because there is actually less O 2 per volume compared to at sea level. Since gases are compressible, this creates an environment in the mountains that has less air per unit volume. The pressure p in pascal (Pa) is equal to the pressure p in millimeter mercury (0°c) (mmHg) times 133.32237, that conversion formula: p(Pa) p(mmHg) × 133.
#747 mmhg to atm how to
How to convert Millimeter Mercury (0☌) to Pascal 1 millimeter mercury (mmHg) is equal to 133.32237 pascal (Pa).

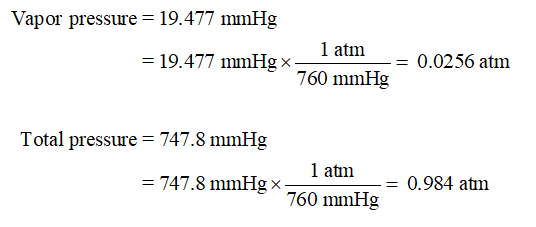
In the mountains there is less air above you so less pressure. 747 Millimeter Mercury (0☌) 99591.80921 Pascal. Simply select the input unit, enter the value and click 'Convert' button. This is simple to use online converter of weights and measures. The same can be said about decreasing pressure. 1 Inches of water 249.08 Pascals: 10 Inches of water 2490.82 Pascals: 2500 Inches of water 622705.02 Pascals: 2 Inches of water 498.16 Pascals: 20 Inches of water 4981.64 Pascals: 5000 Inches of water 1245410.04 Pascals: 3 Inches of water 747.25 Pascals: 30 Inches of water 7472.46 Pascals: 10000 Inches of water 2490820. The pressure value 747.061435973 mmHg (mm of mercury) in words is 'seven hundred and fourty-seven point zero six one four three five nine seven three mmHg (mm of mercury)'. The Diagram below, the pressure at point "X" increases as the weight of the air above it increases. "Atmospheric Pressure" is defined as the force per unit area exerted against a surface by the weight of the air above that surface. The following is a list of all of the standard pressure in every unit for pressure.ġ Atm = 760 torr =760 mmHg = 101.325kPa = 101,325 Pa = 14.7lb/in 2 = 29.92 inHg The SI unit for pressure is the pascal (Pa), but other pressure terms include atmospheres (atms), millimeters of mercury (mmHg), and torr. This is closest to the Moller Massive are gone, so it's likely the identity of the gasses are gone.Gas pressure is a gauge of the number and force of collisions between gas particles and the walls of the container that holds them. And we get a Moeller Mass of 39.8 g per mole. To get the pressure in atmospheres, divide by 760 (note that this is an exact conversion factor) to get 56.3/760 0.0740 8 atm. So to calculate, Mueller Mass will take the mass divided by the moles that correspond to that mass. First, the pressure in mm Hg is just 56.3. All the units cancel except moles, and we get 2.59 times 10 to the negative three moles.
#747 mmhg to atm plus
Well, then take the volume 7 63.8 mL and divide by 1000 to get leaders well, then divide that by our, which is 0.8 to 06 leader atmospheres per Kelvin mole and then multiplied by the temperature in Kelvin, which would be the 22 plus 2 73 to convert the Celsius temperature into Calvin. 47 millimeters of mercury and converted into atmospheres by dividing by 7 60. But we know enough to use the ideal gas law to calculate the moles, which would be in we'll take the pressure 74. Mueller masses the mass of the gas which we know divided by the moles of the gas which we don't. To determine the identity of the gas, we could calculate Mueller Mass. This is done in order to convert the pressure from mmHg to atm, because the value for R contains atm as the pressure unit.

And the difference is the mass of the gas, which was given to us as 0.103 g. P x V n x R x T where R 0.0821 (L atm)/ (mol K) or R 62.4 (L mm Hg) / (K mol) Example: How many moles of air are there in a 2. n (750.0 mmHg / 760.0 mmHg atm¯ 1) (0.890 L) / (0.08206 L atm mol¯ 1 K¯ 1) (294.0 K) Please note the division of 750 by 760. The density of a noble gas is 2.71 g/L at 3.00 atm and 0 ☌. Find the density of Freon11 (CFCl3) at 120 ☌ and 1.5 atm 20. We then measure the mass of the flask when it's evacuated and the mass of the flask after adding the gas. When an evacuated 63.8 mL glass bulb is filled with a gas at 22 ☌ and 747 mmHg, the bulb gains 0.103 g in mass. When the gas is added at 22 degrees Celsius, we end up creating a pressure of 7. Here we have a gas that is used to feel and evacuated glass bulb with the volume of 63.8 mL.
